Mouse Lab Behaviour: How to Listen to Their Preference During Maintenance in House Facilities
ABSTRACT
Mouse phylogeny, evolution and natural behaviour enable a new methodology for testing inputs, materials and equipment that are adequate/comfortable for the animals during their maintenance in house facilities. We developed an ICS prototype, capable of comparing two types of objects/materials based on “listening” the animal. The efficiency of the ICS can be demonstrated through the evaluation of basic materials in house facilities, such as chow, water decontamination process, floor/bed type and environmental enrichment objects. Our results demonstrate that while infant and young mice prefer commercial unprocessed chow over autoclaved one, adult mice show no preferences. Regarding water consume, at any age (4, 6 and 8 weeks of life - wko), differences in the water decontamination process - filtered or autoclaved – were observed. However, in relation to the floor/bed, at all ages, wood shavings were preferred (90%) over pine flakes. Finally, the environmental enrichment categories preferred are those that offer shelter and nesting possibility. Our results showed that ICS uses adequate evaluation parameters, allowing the handler to “hear” the mouse’s preference, and through screening tests insert material/object(s) in its routine that significantly minimize distress and suffering.
KEYWORDS
Animal welfare; Individual and social mouse behaviour; Interconnected cage system
INTRODUCTION
How to raise, maintain and manipulate mice for scientific purposes without compromising their welfare? This is a complex but relevant question in the area of science of laboratory animals and studies that provide subsidies to improve their life quality are welcomed. We believe that the best way to find out how to promote animal’s welfare in house facilities is to “ask” them their preference regarding the procedures, products, equipment and materials that will be used in their maintenance or handling. To answer this question, we need to elaborate a “translator” to the animals, which is the development of technological innovations that enable the communication between laboratory animals and researchers.
Animal Protection & Animal Welfare
With the advance of science, technology and especially medicine (human and veterinary) laboratory animals are important protagonist in the intrinsic chain of knowledge development and of the discovery of new therapies, medicines and vaccines, such as for current situation of the COVID-19 pandemic [1]. Unfortunately, in many cases, in accordance with 3R´s ethical principles, animal replacement is not possible, so reduction of the animal number and the warranty of welfare conditions are underlying requirements for the refinement of animal experiments. Therefore, animal welfare is an important form of animal protection, reducing harm, pain, stress and suffering - during scientific and technological development [2].
Animal Welfare: Concept and Definitions
Animal welfare (AW) can be defined as the state obtained when the animal is able to control internal and external variations and, consequently, remaining in homeostasis or in harmony (balance between the individual and the environment). However, to achieve homeostasis, the animal needs to adapt to adverse situations and control them, so its physiological and behavioural responses are altered, requiring greater metabolic energy [3]. AW can be considered under two different angles: i) absolute AW, described by Hughes [3,4] as being the state of the organism in which there is physical and mental harmony and ii) relative AW, the state of a given organism during its attempts to adjust to its environment, as described by Broom in 1996 [4]. AW scientific assessment is a key element in the implementation of good practices in the use of animals for educational and scientific purposes [5]. The AW involves multiple criteria, such as, identifying opportunities for reduction and refinement intervention as analgesia and noninvasive parameters in handling lab animals [6]. AW analysis must be done with the effective participation of all the employees involved, in a process that tries to understand the perceptions and practical experiences of the manipulators [3,6].
Mouse Lab and Welfare
As described by some authors, in order to offer welfare to the mouse, it is necessary to know its phylogeny, genealogy and ethology (natural and in a house facilities). The mouse is a species that appeared in the region, currently, between Pakistan and India about 14 million years ago. From the fossil of a molar tooth, its origin comes from a common ancestor Artemus chinensis.
During evolution and natural selection promoted the division into several genres, such as Mus, object of our study. In turn, over time, the genus Mus was subdivided into subgenera and species.
The main species are: Mus mus musculus, Mus mus casteneus and Mus mus domesticus. They have a widespread geographical distribution and always maintaining a commensal relationship with humans [7]. In the early twentieth century, some breeders captured wild mice and reproduced them in captivity. Also, at that time, a targeted selection of matings and beginning their introduction into laboratories for scientific studies, mainly albino mice, in the area of genetics and oncology [7-8]; (Figure 1).
Genealogically, after years of laboratory animal science studies mainly based on their artificial selection, including isogenic and transgenic mice, it was suggest that the species that are found today in-house facilities can be genetically described as Mus mus laboratories [8]. One of the main characteristics of the mouse lab is its adaptability. So, the welfare of the mice is closely linked to the knowledge of their behavioural repertoire and their natural instinct. In summary, the basic needs for the comfort of mice in vivarium are: i) ease of food supply; ii) possibility of mating and iii) safe place to find shelter [9]. In short, this stealthy and fearful bio model is highly capable by nature to survive extreme situations and it is in a permanent state of alert, which for us human beings would be a stress-like condition. We suggest that the disruption of the mouse lab welfare is directly related to its inability to adapt to abrupt changes, mainly in its environmental, housing and social interaction, leading to a state of distress [9,10].
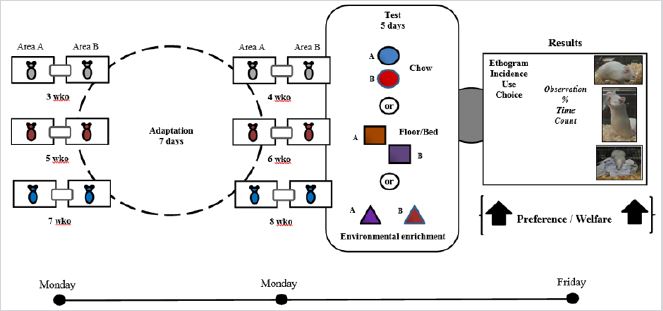
Our research group, for approximately 7 years, has been directly involved in the study of individual and social mice behaviour in house facilities. In the complexity of this theme, such as the diversity of genetic backgrounds (lineages, isogenic and non-isogenic), which in turn promotes a diversity of mice individual and social behaviour, exposure to environmental factors (light, temperature, humidity and etc ...) and space restrictions due to maintenance in the cages in the house facilities. Thus, in this study, we will demonstrate an efficient way to promoted mouse lab welfare is based in the “asking” of their preference. Our proposal is the evaluation of mouse preference through a prototype developed at the LBC/LITEB Animal Laboratory Science Division, the Interconnected Cage System (ICS) (Figure 2), consisting of two cages (independent areas) (Figure 2A), with different inputmaterials and equipment in each cage, linked by a pass-through connection [11]; (Figure 2B). The primordial objective of ICS is the interaction of the animal with different inputs to determine its preferences, promoting greater comfort and welfare. Specifically, in the present work, we will ascertain the preferences of male Swiss Webster mice, at different ages, between types of chow, water processing, floor/beds and environmental enrichment materials.
MATERIALS AND METHODS
Interconnected Cage System (ICS)
The ICS is a prototype structured from the basic concept of allowing the mouse to have two possibilities of choice, demonstrating its preference. It consists of two polysulfone cages (12 x 15 x 30 cm, total area of 360 cm2) autoclavable with a longlasting stainless-steel wire cover, water supplied in autoclavable drinkers (250 ml) and a commercial pelleted chow for rodents. The cages are interconnected by a polyvinyl chloride (PVC) connexion (25 x 3/4”) with flange and decontaminated with 1% sodium hypochlorite solution [12]. The floor/bed is basically lined with original wood shavings, sieved and sterilized (50 g/cage).
Animal and Environmental Conditions
We used 80 male Swiss Webster Stock mice from the Instituto de Ciência e Tecnologia em Biomodelos (ICTB) (FIOCRUZ) in three sets of age, originally received with 3, 5 and 7 weeks old (wko). The animals were adapted to the environmental conditions and ICS for 7 consecutive days. The animals and the ICS were maintained at the experimental house facilities of the LBC/LITEB (Instituto Oswaldo Cruz) in ventilated shelves, with indication of filter saturation and temperature control (20.0±2.0 oC), doors in crystal polycarbonate with magnetic closure and sound insulation. The air exchange inside the shelf was 20 to 25 changes/h and the humidity maintained between 45-55%, with 12-hour light/dark cycles. So, at the beginning of the experiments, after adaptation, the age groups were: infant (4 wko), young (6 wko) and adult (8 wko). For each age, two experiments were performed – trials #1 and #2 - each with 5 individuals per ICS.
Experimental Design
Our objective is to assess the animal’s preference between two different inputs and materials located in the areas A and B of the ICS after a 7-day adaptation. During five days (Monday to Friday) different parameters are evaluated - chow, water, floor/bed and environmental enrichment in the three groups of mice, 4, 6 and 8 wko (Figure 3).
Parameters and Animal Preference
Chow: The animals received a closed commercial formula under two presentations:
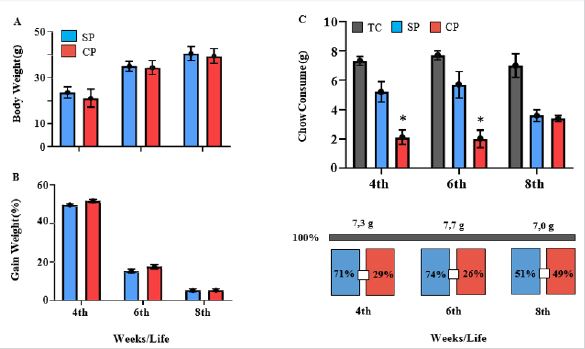
i) non-autoclavable pelleted chow (NC) and ii) autoclavable pelleted chow (AC). The autoclaving process consisted of placing the packaged feed in a vertical autoclave (Phoenix-Brazil, 300 L) at 121 °C/20 min. In trial #1, NA (300 g) was offered in area A, and AC (300 g), in area B, while in trial #2, the reverse occurred, AC in area A, and NA in area B. The animals were weighted at the arrival in the laboratory (7-day adaptation), in the first (initial, Monday) and the fifth (final, Friday) days of the experiments and the individual body weight and the gain weight were calculated. To quantify the daily individual consume of a given chow (300 g in each area), the remaining chow in each area was weighted at the end of experiment, and the difference (consume) divided by the number of animals and the number of days, allowing the determination of the percentual distribution of AC and NC – preference - in relation to the total chow consume (TC).
Water: The interference of the water decontamination process was assessed using
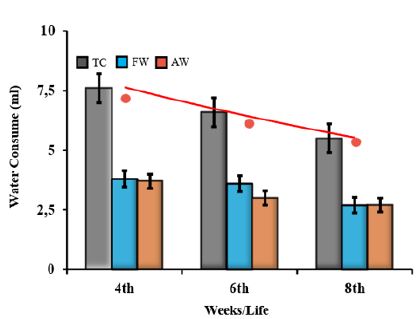
i) filtered water (FW) in a conventional water cooler and ii) autoclaved water (AW) (121 °C/20 min). Likewise, in trial #1, FW (250 ml) was offered in area A and AW (250 ml), in area B, and the reverse occurred in trial #2. The amount of FW and AW drank by each mouse was estimated, by the difference between the initial and final water volume divided by the number of days and the number of animals. The percent of each water type in relation to the total consume (TC) expresses the animal´s preference.
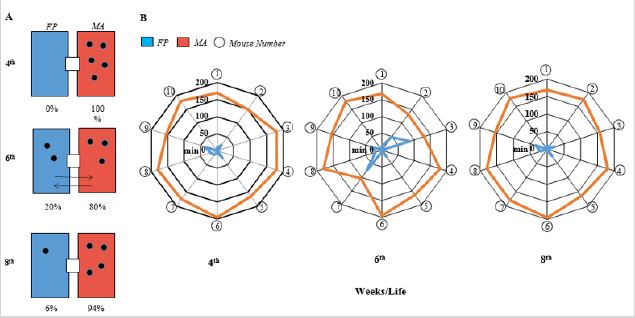
Floor/bed: Two types were used: i) Pine Flakes (PF) and ii) Wood Shavings (WS) produced by RG Granja® (São Paulo, Brazil). Both materials were autoclaved at 121 °C/20 min, and already during the adaptation period, (PF) (100 g) was added to area A or B and (WS) (50 g), to area B of A. Similarly, to the chow and water experiments, in trial #1, PF was added to area A and WS, to area B, and the reverse occurred in trial #2.
The average and percentage values related to the floor/bed preference were determined by the ethogram, by counting the incidence/presence and the length of stay in the areas A or B in a period of 5 days.
a) Ethogram: Through a camera (Sony Shot Hd 6.5®) placed on the ventilated shelves, the ICS was filmed daily for 10 consecutive hours. This footage was used to analyse the ethogram performance based on the main individual and social behaviours of mice: environment exploration, food search, self-hygiene, social interaction and rest.
b) Animals’ incidence and presence in area A or B: During the 5 consecutive days, the number of animals in each area was quantified, in three moments: morning (10:00 PM), afternoon (2:00 PM) and night (6:00 PM). In this way, we were able to calculate the average number of animals that was found during the test period in each area and calculate the total incidence and preference of the animals.
c) Individual permanency time: Through the records, we determined in 60-minute periods: i) presence of animals identified in each area (individual number/area) and ii) time spent in each area (individual time/area). For each age group, 4, 6 and 8 wko, individual counts (5 animals/trial) were performed simultaneously at each collection periods, morning, afternoon and night, totalizing daily 180 min of register.
Environmental enrichment: After a pre-selection carried out by Martins 2018 the assessment of the preferred environmental enrichment was delineated by two categories: i) equipment that provides shelter and ii) nesting materials. Thus, for the shelter category, we compared Igloo® (Alesco Inds, Brazil) versus a PVC tube (10 x 3 cm with diameter of 10 cm), and for the nesting category, we compared two pieces of the crumpled absorbent paper versus a surgical polyester cap with pleated ends and 30 cm of thickness. Our analysis was similar to that used for the floor/ bed preference, ethogram, use/interaction time evaluated for each equipment/material [12]. The results were expressed by the percentage of use of a given object and calculating the preference index (IP), through the formula:
IPset ages = (Number of Interactions x Time of Interaction)/Total Time of Analysis
We would like to emphasize that the experimental design was carried out twice, with inversion in the ICS position. In the first trial a given input/material or equipment was placed in area A, in the second trial, it was placed in area B, to provide more reliable data.
Statistical Analysis
Statistical analyses were performed using the GraphPad Prism program, version 5.0 (Graph-Pad Software Inc.), calculating the group’s mean value and ± SD. In addition, we applied the One-Way ANOVA test. The statistical significance was confirmed by the posttest Turkey, considering a significant difference when p ≤ 0.05.
RESULTS
Our results clearly demonstrate the efficiency of ICS. This system was able to measure the animal’s preference among different types of inputs/materials and equipment that provide them a greater welfare.
Body weight and chow´s Preference: In relation to the ICS feed supply, the parameters: i) Body weight and ii) Weight gain were initially evaluated (Figure 4). As abreviações estão diferentes na figura The individual body weight was calculated for each age group (4, 6 and 8 wko) at the end of each trial, and no difference in the body weight was observed between non-autoclaved and autoclaved chows - NC and AC. The values (NC versus AC), in g, were for the infant group (4 wko) 24.6±2.9 versus AC: 24.2±2.4, for the young (6 wko) 39.3±3.2 versus 38.5±2.6 and for the adult (8 wko), 44.6±4.2 versus 44.2±4.2 (Figure 4A). As expected, the weekly gain weight was similar for NC and AC, being higher from 3 to 4 wko (NC: 49.6% and AC: 51.6%) than from 5 to 6 wko (NC: 15.2% and AC: 17.4%) and 7 to 8 wko (NC: 5.1% and AC: 5.2%) (Figure 4B). When animals chow´s preference was evaluated by the individual consume, the total consume was similar for all ages, infants (7.3±0.4 g), young (7.7±0.6 g) and adults 7.0±0.3 g). However, a preference for the non-autoclaved chow (NC) over AC was observed in the infant and young groups: 71.0 x 29.0% (4 wko), and 74 x 26% (6 wko). In adult mice (8 wko) no significant differences were observed between the NC (51%) and the AC (49%) (Figure 4C).
Water´s Preference
The water palatability directly interferes with the animal’s intake volume and compromises its welfare. In this way, ICS was used to compare the animals’ preference for the water supply – filtered (FW) or autoclavable (AW) (Figure 5). First, our results showed a decrease of the individual total water consumption (TC from infancy to adulthood, 7.5±1.0 ml (4 wko), 6.6±0.6 ml (6 wko) and 5.5±0.5 ml (8 wko). Regarding the mice water decontamination process, no significant difference in the preference for water intake was observed. The values (FW versus AW), in ml, were for the infant group (4 wko) 3.8±0.3 versus 3.7±0.4, for the young (6 wko) 3.6±0.2 versus 3.0±0.2 and for the adult (8 wko), the values were the same, 2.7±0.1 ml (Figure 5).
Floor/Bed Preference
The floor/bed used directly affects animal´s welfare, in our case Pine Flakes (PF) and Wood Shavings (WS) (Figure 6). In trial #1 the number of animals in the respective areas (A or B) were determined at three different periods of the day (morning, afternoon and night). For infant mice (4 wko) the preference was 100% for WS, while for young (6 wko) and adults (8 wko) a higher activity and interaction within ICS was observed with increased changes between areas A and B. At 6 wko, WS preference was maintained, 80%, with 20% in the PF floor area. At 8 wko, preferences 94% for WS and 6% for PF (p≤0,05) (Figure 6A). In trial #2, despite individual heterogeneity, the results corroborate the mice preference for WS. In relation to the individual permanency, at 4 wko the mice stayed 173.0±9.8 min at the WS area and 9.0±11.3 min at the PF area (p≤0.05), young animals (6 wko), 154.5±26.1 min in WS and 25.5±26.1 min in PF (p≤0.05); for adults (8 wko), the corresponding values were 170.5±11.7 min and 9.9±11.7 min (p≤0.05). The ethogram results showed individual Flor Bed interaction and social behaviour by ICS use. Observed that infant animals have rest, self-cleaning, food consume, nesting and physical contact directly related with WS offer. Young animals are extremely curious and demonstrated high intensity of two area use by use the cage. Can be observed PF area was mice used to excretion and WS social contact, nesting and resting. At 8th wko, occurs individual exploratory activity decrease, however, mouse adults showed eat activity in the PF, but, again, social contact and rest is performed in WS floor bed.
Environmental Enrichment Preference
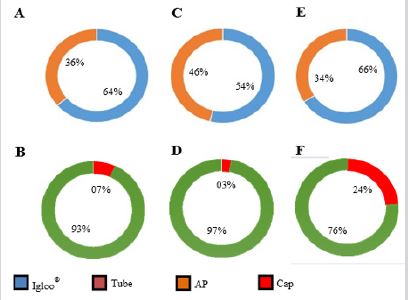
For the three age groups, the preferences of male Swiss Webster mice for shelter and nesting materials were evaluated (Figure 7,8). At 4 wko, the mice displayed a preference for the Igloo® (64%) when compared to the PVC tube (36%) (p≤0.,05) (Figure 7A), and for Absorbent Paper (AP) (93%) when compared to Surgical Cap (SC) (7%) (p≤0.05) (Figure 7B). At 6 wko, the corresponding values for shelter were 54% for Igloo® and 46% for the PVC tube (Figure 7C), and for nesting, 97% for AP versus 3% for SC (p≤0.05) (Figure 7D). In adulthood (8 wko) the mice showed similar results, 66% Igloo® versus 34% PVC (Figure 7E) and 76% for AP versus 24% for SC (p≤0.05) (Figure 7F). Incidence and presence were assessed in different forms. The incidence was determined by the use percentage of the object and the presence by the usage time that the animal interacted with object.
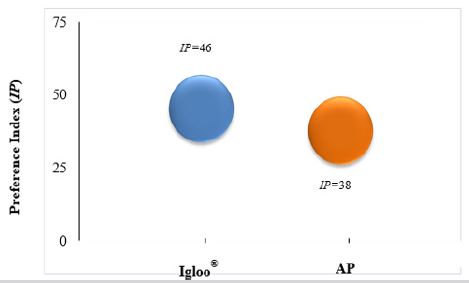
Thus, the preference index (IP) for Igloo® and for AP were calculated (Figure 8). Was observed in all set of ages, a subtle difference between Igloo® and AP as environmental enrichment. Therefore, PVC and SC were not effective equipment/materials to promote mice environmental enrichment in house facilities. Finally, during the ethogram analysis, for the 4 wko group, since rest is the preponderant behaviour, objects that encourage nesting are recommended. Moreover, for these infant mice, nest “construction” ability is very low. At 6 wko, there is a high interaction with AP and a higher nesting ability and interaction. The nest material suffers a fast wear (24 to 48 h), becoming part of the floor; and in relation to shelter objects a higher interaction and curiosity was observed with Igloo over AP. At 8 wko, the physical activity decreases and, despite an increased interaction with AP, the higher interaction is still with Igloo®. Then, IP value determines the relation between the animal´s interaction count (number of events) and the interaction time with environmental enrichment object. In other words, their preference.
DISCUSSION
Currently we can observe that the study in the area of Science of Laboratory Animals is not always taken in account. Through a biometric analysis in the site National Center for Biotechnology Information, U.S. National Library of Medicine, in the PubMed module (https://www.ncbi.nlm.nih.gov/pubmed/), the association of the key words “Mouse” AND “Welfare” showed that only 2.4% of the publications are related to mouse biomodel. These biometric results: i) indicate the need for an increase in the study of the refinement and well-being of mice and ii) allows an anthropocentric interpretation in relation to the means of promoting animal welfare [13].
The Interconnected Cage System (ICS) allows to evaluate male Swiss Webster Outbred Stock preferences, at different ages, infant, young and adult [12]. So, what could be considered a detail, we consider ourselves extremely relevant: evaluate, or even, “ask” the animal what would be his preference (choice) in relation to inputs, materials and equipment in house facilities. Intrinsically, the mouse is an animal prepared for stressful situations. So, the issue becomes more complex, because naturally we would be working with a limitstress model [14-16]. Moreover, the main biomarker for assessing WA impairment is the serum dosage of corticosterone, but the procedure induces discomfort and stress [17]. Paradoxically, corticosterone increased levels are a natural part of the adaptation process to a stressor factor. So, we suggest that only in two situations corticosterone levels can define AW impairment: i) low levels of the hormone when the animals are under a stressful situation and ii) maintenance of high corticosterone levels for long periods [18,19].
Thus, this study proposes that the choice itself, the animal preference through the ICS, is the best way to offer comfort, life quality and WA during maintenance in house facilities. Our results clearly demonstrate the efficiency of the new ICS methodology enable mouse preference. We must point out that mouse phylogeny and evolution promote in murine species a stress-like physiological system comparable to that in humans [7,8]. In addition, according to Hughes’ definition of relative welfare, it is closely linked to the animal’s ability to adapt to stressful situations, especially when promoted by changing its routine. [4]. We emphasize that mouse lab has a remarkable characteristic of adaptability to new environments, food supply and its individual and social behavioural complex repertoire. Thus, the relationship between serum corticosterone and distress is only reliable if there is a permanent increase in the dosage of this hormone, since corticosterone promotes negative feedback in stress responses [18,19].
At present moment, non-invasive parameters to evaluate AW are subjective, such as, the study of the relation with facial expressions and emotional state through Mouse Grimace Scale (MGS) [19,20]. Moreover, assays involving noxious stimuli of moderate duration are accompanied by facial expressions of pain [21,22].
Technological innovation and the relevance of our methodology consists of a non- invasive evaluation procedure before inserting new materials/objects and procedures in the animals’ handling routine, allowing AW, and thus, not compromising the reproducibility of the assays. In relation to the chow supply, we observed that probably the palatability of unprocessed chow is bigger, being the preferred food for infant and young mice [23]. Since to preserve sanitary barriers and health status, chow sterilization is needed, based on products currently offered on the market, irradiated commercial chow could be indicated. Regarding water, the results demonstrated that the autoclaving process does not interfere with animal consume [24]. A critical point is the discussion about the best floor/bed [25]. A rational choice is for a more absorbent material, leading to decreased ammonia level and number of exchanges (cages), [25]. Our results showed the unquestionable mice preference for the WS when compared to the PF. We believe that this fact is linked to the interaction animal-material, such as individual behavioural attitudes as: i) exploration and search environments and ii) comfortable nesting for rest [26]. Finally, in relation to environmental enrichment, our study demonstrated that the animal’s preference is for equipment that allows shelter [12]. The main environmental enrichment objects chosen have advantages and disadvantages. The shelter (Igloo®), promotes place dominance dispute with aggressive behaviour, however it is easily decontaminated. Absorbent Paper, on the other hand, needs specific decontamination protocol before use in the cage, and must be changed every 48 h, however it stimulates social interaction and physical contact. We emphasize that from the moment the environmental enrichment program is implemented, it should not be interrupted, as it causes an anxious-like state in the animals in the absence of the equipment/materials [27,28].
CONCLUSION
The main conclusion that we achieved in this work is the effectiveness of the ICS prototype in providing AW mice at the house facilities. Thus, in accordance with ethical principles, current legislation and normative resolutions aiming an AW status. The application of the ICS tests allows a choice by the animal and use of preferred material or object promotes AW. The originality of the ICS consists of “listening” to the animal’s preference when different inputs, materials, equipment and procedures are compared, encouraging further studies about AW and mice.
We believe that regulation, standardization and the few studies regarding the AW preservation of mice in animal houses allow the choice of inputs, materials and equipment based on an anthropocentric perception. In this way, the ICS makes it possible to choose the mouse, thus making it more reliable in raising the animals’ quality of life.
ACKNOWLEDGMENT
Dr. Solange Lisboa de Castro for the critical reading of the manuscript; Post-graduation program of Ciência de Animais de Laboratório from the Instituto de Ciência e Tecnologia em Biomodelos (ICB)/FIOCRUZ; Laboratório de Biologia Celular and Instituto Oswaldo Cruz (IOC). The procedures were approved by the Comissão de Ética no Uso de Animais de Laboratório (CEUAIOC) under the license No L- 004/2016.
REFERENCES
- Oliveira GM (2020) Laboratory mouse & COVID-19 research. Open Acc J Bio Sci 2(2): 338-340.
- Oliveira GM (2019) Use of laboratory animals in a pre-clinical trial: A critical view of the bio models development and application. Am J Biomed Sci & Res 4(2): 152-154.
- Rivera E (2017) Bem-Estar de Animais de Laboratório. In: Lapchik VV, Mattaraia VM, Ko GM (Orgs.). Cuidados e manejos de animais de laboratório. 2nd (edn.). Rio de Janeiro: Ateneu, pp: 35-45.
- Broom DM (2011) History of animal welfare science. Acta Biotheoretica 59(2): 121-137.
- Broom DM (1996) Animal welfare defined in terms of attempts to cope with the environment. Acta Agriculturae Scandinavica 27: 22-28.
- Appleby MC, Hughes BO, Mench JA, Olsson A (2011) Animal welfare, 2nd (edn.). Wallingford, England.
- da Costa SM, Rossi MID, Evagelista AA, Oliveira GM (2019) Origin, phylogeny and natural behavior of mice: what is their influence on welfare during their maintenance in the house facilities? Am J Biomed Sci & Res 5(5): 356-361.
- Evangelista AA, da Costa SM, Rossi MID, Evagelista AA, Oliveira GM (2019) Wild mouse & laboratory mouse historical aspects, genetic selection and welfare. R Soc bras Ci Anim Lab 7: 122-129.
- Oliveira GM (2012) Hierarquização em Agrupamentos. In: Mattaraia VM, Oliveira GM (Orgs.). Comportamento de Camundongos em Biotério. São Paulo: Poloprint 179-194.
- Kuzel MA, da Costa SM, Evagelista AA, Oliveira GM (2013) Estudo da hierarquia de camundongos Swiss Webster através do uso de sistemas com gaiolas interligadas (SGI). R Soc bras Ci Anim Lab 2: 49-60.
- Oliveira GM, Goncalves MAB, Veronez TA (2018) Enriquecimento Ambiental: Qual a melhor forma de utilização de enriquecimento ambiental para camundongos em biotério? 1st Edn. Ebook. São Paulo, Brazil 01-119.
- Furtado AK, Oliveira GM (2018) Análise biométrica relacionada an importância do bem-estar de camundongos e an influência nos resultados dos ensaios científicos. R Soc bras Ci Anim Lab 6: 111-128.
- Gilmore AJ, Billing RL, Einstein R (2008) The effects on heart rate and temperature of mice and vas deferens responses to noradrenaline when their cage mates are subjected to daily restraint stress. Laboratory Animals 42(2):140-148.
- Haller M, Mironov SL, Richter DW (2001) Intrinsic optical signals in respiratory brain stem regions of mice: neurotransmitters, neuromodulators and metabolic stress. J Neurophysiol 86(1): 412-421.
- Reis-Silva TM, Sandini TM, Calefi AS, Orlando BC, Moreira N, et al. (2019) Stress resilience evidenced by grooming behaviour and dopamine levels in male mice selected for high and low immobility using the tail suspension test. Eur J Neurosci 50(6): 2942-2954.
- Retana-Márquez S, Bonilla-Jaime H, Vazquez-Palacios G, Martínez-Garcia R, Velázquez-Moctezuma, J (2003) Changes in masculine sexual behavior, corticosterone and testosterone in response to acute and chronic stress in male rats. Horm Behav 44(4): 327-337.
- Reiter CP, Christy AC, Olsen CH, Bentzel DE (2017) Effect of home cage bedding in the induction chamber on serum cortisol and corticosterone levels in response to isoflurane-induced anaesthesia in C57Bl/6J mice. J Am Assoc Lab Anim Sci 56: 118-121.
- Oliveira FS, Evagelista AA, Oliveira GM (2015) Dosagem da concentração plasmática de corticosterona em camundongos altamente agressivos da linhagem Swiss Webster. R Soc bras Ci Anim Lab 3: 85-94.
- Tuttle AH, Molinarto AJ, Jethwa JF, Sotocinal SG, Prieto JC (2018) A deep neural network to assess spontaneous pain from mouse facial expressions. Mol Pain 14: 1744806918763658.
- Langford DJ, Bailey AL, Chanda ML (2010) Coding of facial expressions of pain in the laboratory mouse. Nat Methods 7: 447-449.
- Konstandi M, Johnson E, Lang MM, Marselos M (2000) Noradrenaline, dopamine, serotonin: different effects of psychological stress on brain biogenic amines in mice and rats. Pharmacol Res 41(3): 341-346.
- Lapchik VV, Mattaraia VM, Ko GM (2017) Cuidados e manejos de animais de laboratório. 2nd edn. Rio de Janeiro: Ateneu 11-27.
- Rosenbaum MD, Vandewoude S, Johnson TE (2010) Disparities in ammonia, temperature, humidity, and airborne particulate matter between the micro-and macroenvironments of mice in individually ventilated caging. Journal of the American Association for Laboratory Animal Science 49: 177-183.
- Rosenbaum MD, Vandewoude S, Johnson TE (2009) Effects of cage-change frequency and bedding volume on mice and their microenvironment. J Am Assoc Lab Anim Sci 48: 763-773.
- Spangenberg EM, Keeling LJ (2015) Assessing the welfare of laboratory mice in their home environment using animal-based measures a benchmarking tool. Laboratory Animals 50(1): 30-38.
- Yildirim F, Yildirim BA, Yildiz A, Terim KA, Cengiz S, et al. (2017) Evaluation of perlite, wood shavings and corncobs for bedding material in rats. J South African Vet Assoc 88: 130-137.
- Fabrício VL, Oliveira AS, Lapchik VV (2014) Benefícios sanitários do sabugo de milho enriquecido ou não para roedores e técnicos de laboratório. R Soc bras Ci Anim Lab 2: 49-56.
- Oliveira GM, Brito CF (2019) Influence of social isolation and aggressive behaviour in the appearance of depression-like in experimental model. Arch Depress Anxiety 5: 042-046.
Article Type
Research Article
Publication history
Received date: August 01, 2020
Published date: August 21, 2020
Address for correspondence
Gabriel Oliveira, Laboratório de Biologia Celular, Instituto Oswaldo Cruz, Fundação Oswaldo Cruz, Brazil
Copyright
©2020 Open Access Journal of Biomedical Science, All rights reserved. No part of this content may be reproduced or transmitted in any form or by any means as per the standard guidelines of fair use. Open Access Journal of Biomedical Science is licensed under a Creative Commons Attribution 4.0 International License
How to cite this article
Thaís V, Miguel B, Maria IDR, Gabriel O. Mouse Lab Behaviour: How to Listen to Their Preference During Maintenance in House Facilities. 2020 - 2(4) OAJBS.ID.000208.
Figure 1: The origin of the mouse in the Asian region (A) was determined by its original ascendant, through the teeth molar fossil (in detail) after dinosaurs’ extinction and appearance of eutheric species. The most common species are Mus mus musculus (B) and Mus mus domesticus (C) that gave rise to Mus mus laboratorius (D) used for scientific purposes.
Figure 2: The prototype of the interconnected cage system (ICS) is based on the principle of giving the mouse access to two different areas (A). From this premise, a mice group, at different ages (in separate systems) are able to demonstrate its choice (preference) between different types of objects placed in the two areas. An example was the placing of environmental enrichment objects (ball with rattle - playful activity) in area A and an Igloo® as a shelter in area B. So, through the connection (passage), we can measure the use and time of the interaction to each object and define the mouse´s preference (B).
Figure 3: The scheme illustrates the simplicity and efficiency of ICS application. On Monday, male Swiss Webster mice with 3, 5 and 7 wko received from ICTB were placed in the ICS for one week for adaptation. After that, the next Monday (initial point), the ICS was cleaned and the inputs (chow, water and floor/bed) were inserted and maintained till Friday (final point). The three age groups were analysed simultaneously. Between these five days, the ICS was filmed and parameters such as ethogram, animal´s incidence and permanence (OR presence?) and time of the interaction animals were evaluated.
Figure 4: Evaluation of the decontamination process (autoclaving) of the commercial pelleted chow. Analysis of the body weight (A) and gain weight (B), no significant differences were observed between the consume of chow unprocessed (NC-blue bar) and processed (AC-orange bar). The mice preference in relation to decontamination chow process (TC total consume, grey bar) was evident, the consume of the non-autoclaved chow (NC) was significantly higher than that of AC. Asterisks show statistical significance between NC and AC (p≤0.05).
Figure 5: The water decontamination process does not interfere with animal consumption (ml), it only shows an evident decreased in total consumption (TC-gray bar) between ages (red line). However, the filtration process (FW-blue bar) or the autoclaving use (AW-orange bar) does not interfere in the preference animals’, at any age.
Figure 6: The comparison between the Floor/Bed types shows an evident preference of mice for wood shavings. During all measurement period, at three different times of the day/5 consecutive days, we counted the animals that interacted (A) with the pine flakes (FP-blue box) and the wood shavings (MA - orange box) and calculated the percentage of number of animals use (double trial illustrative) at the ages of 4, 6 and 8th wko. In addition, through film recording, was evaluated 10 individuals total (circles) and time estimated (min) of the mice’s area remains (B) with pine flakes (FP - blue line) and in wood shavings area (MA- orange line) during the infant (4th wko), young (6th wko) and adult (8th wko) ages.
Figure 7: When evaluated equipment/materials for environmental enrichment, the two categories that were most chosen were “shelter” and “nest”. Regarding the objects of preference in these categories, the use percentage showed in 4 (A), 6 (C) and 8th wko (E) a higher prevalence of the Igloo® use (blue fillet) when compared to the Tube (orange fillet). In the case of nesting behavior, infants (B), young (D) and adults (E) used Absorbent Paper (AP - green fillet) more than the surgical cap (Cap - red fillet).
Figure 8: Carrying out the environmental enrichment object selection, using ICS scheme, we compared the mice objects preference at the house facilities, which were Igloo® and Absorbent Paper (AP). This evaluation consisted of the IP formula, which means the relationship between the number and time of animal’s interaction with objects. The shelter category (blue circle), represented by Igloo®, showed a slightly higher preference than AP (green circle) in the average of all ages studied.