Efficacy of Letrozole as Maintenance Therapy for Ovarian Cancer
ABSTRACT
Ovarian cancer is a persistent disease with periods of recurrence. Surgery with subsequent chemotherapy is not sufficient to stop the disease. The aim of this study is to establish whether letrozole applied as maintenance therapy can prolong life without progression. Thanks to exposure of estrogen receptors in ovarian cancer cells, hormonal therapy provides an alternative treatment method. Aromatase inhibitors block synthesis of estrogen and prevent progression of ovarian cancer. More advantages are observed when using aromatase inhibitors (letrozole) in low-stage serous and endometrioid ovarian cancer. However, further investigations are needed to better evaluate this maintenance treatment.
KEYWORDS
Ovarian cancer; Letrozole; Maintenance therapy; Estrogen; Aromatase inhibitor
INTRODUCTION
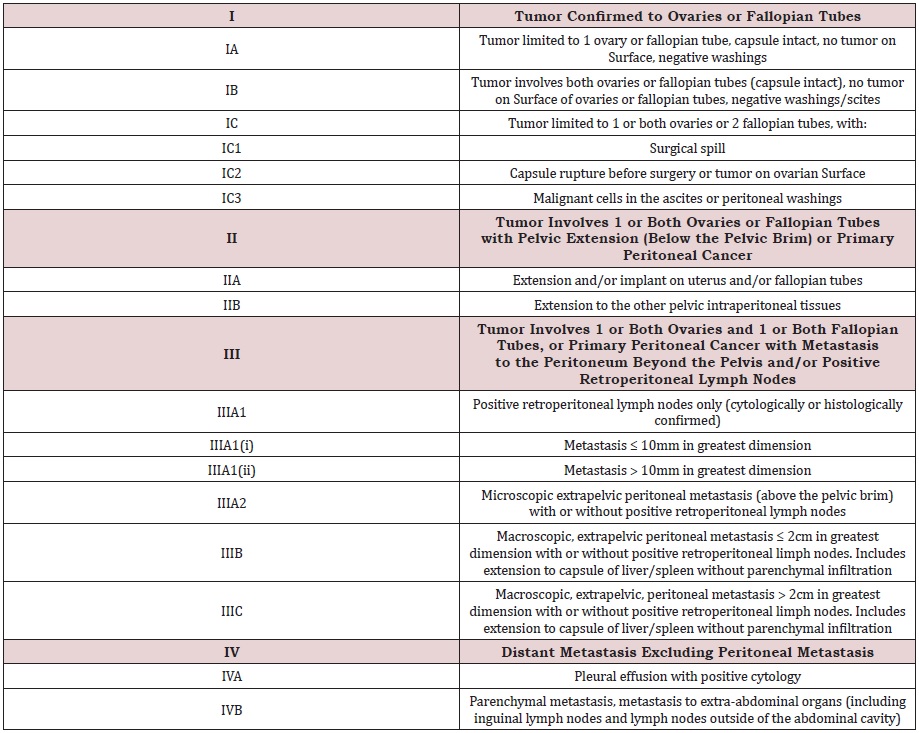
Ovarian cancer is a recurrent disease. Due to unspecific syndromes, the diagnosis is often too late and usually an advanced stage of cancer is revealed. The main choice of treatment is radical surgical treatment followed by chemotherapy. In cases of diagnosis of diffused ovarian cancer, neoadjuvant chemotherapy is the first line of treatment and then surgery is preferred. Special FIGO classification was introduced to assess stage of ovarian cancer (Table 1). Moreover, estrogen receptors, especially ERα, are expressed in more than 80% of high and low grade serous, endometroid ovarian cancer and granulosa cell tumors [1]. It is observed that estrogen receptors are presented in 38-60% of all ovarian cancers, of which 80% are endometrial histological types [2]. This can be a target of a new method of therapy.
The idea of hormonotherapy in recurrent ovarian cancer is based on the estrogen hypothesis connected with exposure of ovarian surface epithelium to estrogen. Additionally, fat tissue containing cholesterol is converted into estrogens, increasing the level of endogenous estrogens. This mechanism is indicated by overweight and obesity as a risk factor of hyperestrogenism and its consequences. In ovarian cancer estrogens receptors are positive in 36% of cases. Therapy based on aromatase inhibitors block synthesis of estrogens and exhibit antitumor effects against ovarian cancer [3]. According to the National Comprehensive Cancer Network guidelines (version 2.2021) hormonotherapy can be a variant of maintenance therapy of ovarian cancer. Aromatase inhibitors and tamoxifen are allowed for low-grade ovarian cancer and endometrioid type of OC.
The aim of the study was to compare the benefits and disadvantages of letrozole as a maintenance therapy of ovarian cancer.
DISCUSSION
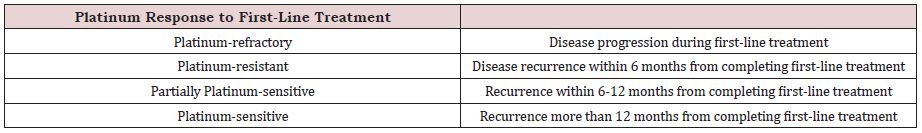
The main question is why it is a good option of treatment. In Cunat et al. [4] discovered higher expression of aromatase in normal ovarian tissue than in cancer tissue and detected aromatase in epithelium. Aromatase P450 is also present inside the endometrioid tissue with higher estrogen expression. Thanks to this, there is a potential role of aromatase inhibitors in treatment of endometriosis [5]. A multi-center phase II study investigated letrozole as one of the aromatase inhibitors in treatment of recurrent endometrioid endometrial cancer with clinical benefits in 50% of the analyzed population [6]. Antiestrogen therapy is an alternative way of treatment. This is not a routine management. For better analysis, different studies documenting the influence on different histological types of ovarian cancer are presented. It is also significant information about platinum response on treatment (Table 2).
Estrogen Receptor-Positive Ovarian Cancer
The first study on the analysis of estrogen receptor-positive ovarian cancer was conducted in 2007 by Smyth et al. It was a phase II study. Each patient took 2.5mg of letrozole orally daily. In all case estrogen receptors in ovarian cancer were observed. Analysis based on 42 women revealed 17% of patients with more than 50% of regression of the disease. No progression was observed in 26% of cases following 6 months of treatment. In 26% of patient’s progression-free survival (PFS) was longer than 6 months. Radiological assessment showed in 9% partial remission and in 42% stabilization of the disease at 12 weeks [7].
Low-Grade Ovarian Cancer
Hormonotherapy in low grade serous ovarian cancer was induced after primary cytoreductive surgery as an adjuvant hormonal monotherapy. Fader et al. published a retrospective study based on 27 patients with stage II-IV of low-stage ovarian cancer after surgery with the majority of women in stage III C (18/27 patients). Histologically, 96% of ovarian cancers were estrogen receptor positive, while 32% showed expression of progesterone receptors. Hormonal monotherapy consisted of use of letrozole (55.5% of cases), anastrozole (37.1%) or tamoxifen (7.4%). After 41 months of observation in 6 cases recurrence occurred and 2 patients died. As a result, it was found that PFS and overall survival were increased in 2-year observation (82.8%; 96.3%) and 3-year follow-up (79.0% and 92.6%) [8].
Gerhenson et al. [9] in 2017 investigated a group of women with stage II to IV low-grade serous carcinoma of the ovary or peritoneum after primary cytoreductive surgery and further platinumbased chemotherapy. The analyzed population was divided into patients with maintenance hormonotherapy and women under observation without additional treatment. The second subgroup consisted of 70 women treated with letrozole (54.3%), anastrozole (2.9%), tamoxifen (28.6%), leuprolide acetate (7.1%) and a small percentage taking a combination of two drugs. Median PFS of the whole group was 32.6 months. For patients under observation median PFS was 26.4 months, while in the second group it was 64.9 months. In the comparison of patients with clinically free ovarian cancer after completion of chemotherapy, hormone therapy gave 81.1 months of PFS, in contrast to 30 months. Women with a partial response to chemotherapy with persistent ovarian cancer had longer PFS (38.1 months) than those under observation (15.2 months). Extended PFS was observed in estrogen-receptor positive ovarian cancer. Of course, patients with stage IV ovarian cancer had a higher risk of progression than those with lower stage disease.
In 2020, Fernandez et al. [10] investigated presence of estrogen and progesterone receptors in low-grade serous ovarian cancer with 56-month follow-up observation and the influence of hormonal treatment. Expression of estrogen receptors was observed in 96% of women, in contrast to 67% observed for progesterone receptors. As a result, higher expression of estrogen receptors in low-grade serous ovarian cancer was correlated with better overall survival. However, hormonal treatment (estrogen and tamoxifen) had no influence on cancer cell proliferation. Another study conducted by Marchetti et al. [11] showed benefits of letrozole in advanced ovarian cancer as treatment of lower toxicity with longer survival without progression, especially in advanced low-stage ovarian cancer. Nica et al. [12] presented analysis based on the financial aspect of hormonal therapy as a maintenance treatment in advanced low grade serous ovarian cancer. In their study, letrozole was confirmed as a cost-effective method of therapy with less toxicity, with the advantages of stable or better quality of life and longer PFS.
High-Grade Ovarian Cancer
Ramirez et al. [13] observed patients with recurrent highgrade platinum and taxane-resistant ovarian and peritoneal cancer treated with a dose of 2.5 mg of letrozole orally. Clinical benefit was observed in 26% of cases. Stable disease was observed in 23%, partial response in 3%, and no complete response was seen. Adverse effects were not severe, with the most frequent being fatigue and diaphoresis.
On the other hand, Papadimitriou et al. [14] conducted a phase II study on women with recurrent epithelial ovarian cancer treated with letrozole as maintenance therapy. Objective response to treatment was observed in 15% of cases. Toxicity of letrozole was generally low. No correlation between response to treatment and estrogen or progesterone expression was found.
Letrozole in Endometrioid Ovarian Cancer
Pan et al. [15] described cases of endometrioid ovarian cancer treated with letrozole. They concluded that the time of induction of antiestrogen therapy is significant. PFS was longer if the maintenance therapy was conducted directly after completion of chemotherapy. Additionally, remission after carboplatin with gemcitabine and letrozole was no longer than 5 months. More advantages were observed in endometrioid cancer with a lower tumor burden after primary chemotherapy.
Letrozole as Part of Combination Therapy
Letrozole has also been used in combination therapy. Colon- Otero et al. [16] conducted a phase II trial with ribociclib and letrozole in endometrial cancer and ovarian cancer with positive estrogen receptors. The analysis showed more advantages for lowgrade serous ovarian cancer (100% of cases of this type) and stage 1 or 2 of endometrioid type in 45.5% of women with this diagnosis. In 50% of estrogen receptor positive ovarian cancer cases PFS was 12 weeks, meeting the endpoint of the study. Definitely less benefit was observed in high-grade serous ovarian cancer. However, one patient (6% of all) was treated for 24 weeks without progression.
On the other hand, letrozole was combined with everolimus in a phase II trial in patients with relapsed estrogen-receptor-positive high-grade ovarian cancer. The daily dose of letrozole was 2.5mg, with everolimus 10 mg per day orally. In 47% of patients promising 12-week PFS with less toxicity was observed [17]. In 2020 Frisone et al. [18] described a case of a patient with serous ovarian cancer with homozygous deletion of the CDKN2A gene with a combination of palbociclib and letrozole as a method of treatment. A good clinical response was observed for 12 months.
CONCLUSION
There have been many studies investigating letrozole as a maintenance therapy in recurrent ovarian cancer. However, the analyzed groups were not sufficiently representative to conclusively support application in a larger population. Letrozole presents more benefits for low-stage serous and endometroid recurrent ovarian cancer. Unfortunately, progression of the cell cancer line to a high grade is often observed, so letrozole alone is not sufficient. Blocking progression of high grade serous ovarian cancer through maintenance therapy requires a further line of chemotherapy. Surely, more investigations are necessary to understand this problem better.
REFERENCES
- Langdon SP, Herrington CS, Hollis RL, Gourley C (2020) Estrogen signaling and its potential as a target for therapy in ovarian cancer. Cancers 12(6): 1647.
- Andersen CL, Sikora MJ, Boisen MM, Ma T, Christie A, et al. (2017) Active estrogen reccceptor-alpha signaling in ovarian cancer models and clinical specimens. Clin Cancer Res 23: 3802-3812.
- Yokoyama Y, Mizunuma H (2013) Recurrent epithelial ovarian cancer and hormone therapy. World J Clin Cases 1(6): 187-190.
- Cunat S, Rabenoelina F, Daurès JP, Katsaros D, Sasano H, et al. (2005) Aromatase expression in ovarian epithelial cancers. J Steroid Biochem Mol Biol 93(1): 15-24.
- Hashim HA (2014) Potential role of aromatase inhibitors in the treatment of endometriosis. Int J Womens Health 6: 671-680.
- Soliman PT, Westin SN, Iglesias DA, Fellman B, Yuan Y, et al. (2020) Everolimus, letrozole and metformin in women with advanced or recurrent endometrioid endometrial cancer: A multi-center, single arm, phase II study. Clin Cancer Res 26(3): 581-587.
- Smyth SJ, Gourley C, Walker G, MacKean M, Stevenson A, et al. (2007) Antiestrogen therapy is active in selected ovarian cancer cases: the use of letrozole in estrogen receptor positive patients. Clin Cancer Res 13(12): 3617-3622.
- Fader A, Bergstrom J, Jernigan A, Tanner E, Roche K, et al. (2017) Primary cytoreductive surgery and adjuvant hormonal monotherapy in women with advanced low-grade serous ovarian carcinoma: Reducing overtreatment without compromising survival? Gynecol Oncol 147(1): 85-91.
- Gershenson D, Bodurka D, Coleman R, Lu K, Malpica A, et al. (2017) Hormonal maintenance therapy for women with low-grade serous cancer of the ovary or peritoneum. J Clin Oncol 35(10): 1103-1111.
- Fernandez ML, Dawson A, Kim H, Lam N, Russell H, et al. (2020) Hormone receptor expression and outcomes in low-grade serous ovarian carcinoma. Gynecol Oncol 157(1): 12-20.
- Marchetti C, Felice F, Ergasti R, Scambia G, Fagotti A (2020) Letrozole in the management of advanced ovarian cancer: an old drug as a new targeted therapy Int J Gynecol Cancer 30(7): 1058-1064.
- Nica A, Lee J, Hong N, May T (2021) Cost-effectiveness of maintenance hormonal therapy in patients with advanced low grade serous ovarian cancer. Gynecol Oncol 160(1): 206-213.
- Ramirez P, Schmeler K, Milam M, Slomovitz B, Smith J, et al. (2008) Efficacy of letrozole in the treatment of recurrent platinum- and taxaneresistant high-grade cancer of the ovary or peritoneum. Gynecol Oncol 110(1): 56-59.
- Papadimitriou CA, Markaki S, Siapkaras J, Vlachos G, Efstathiou E, et al. (2004) Hormonal therapy with letrozole for relapsed epithelial ovarian cancer. Long-term results of a phase II study. Oncology 66(2): 112-117.
- Pan Y, Kao MS (2010) Endometrioid ovarian carcinoma benefits from aromatase inhibitors: case report and literature review. Curr Oncol 17(6): 82-85.
- Colon-Otero G, Zanfagnin V, Hou X, Foster NR, Asmus EJ, et al. (2020) Phase II trial of ribociclib andletrozole in patients with relapsed oestrogen receptor-positive ovarian or endometrial cancers. ESMO Open 5(5): e000926.
- Colon-Otero G, Weroha SJ, Foster N, Haluska P, Hou X, et al. (2017) Phase 2 trial of everolimus and letrozole in relapsed estrogen receptor-positive high-grade ovarian cancers. Gynecol Oncol 146(1): 64-68.
- Frisone D, Charriera M, Clementb S, Christinatb Y, Thouvenina L, et al. (2020) Durable response to palbociclib and letrozole in ovarian cancer with CDKN2A loss. Cancer Biol Ther 21(3): 197-202.
Article Type
Review Article
Publication history
Received date: September 01, 2021
Published date: September 21, 2021
Address for correspondence
Magdalena Bizoń-Szpernalowska, Chair and Department of Obstetrics, Gynecology and Gynecological Oncology, Medical University of Warsaw, Poland
Copyright
©2021 Open Access Journal of Biomedical Science, All rights reserved. No part of this content may be reproduced or transmitted in any form or by any means as per the standard guidelines of fair use. Open Access Journal of Biomedical Science is licensed under a Creative Commons Attribution 4.0 International License
How to cite this article
Bizoń-Szpernalowska M, Sawicki W. Efficacy of Letrozole as Maintenance Therapy for Ovarian Cancer. 2021- 3(5) OAJBS.ID.000320.