Comparison of Tocilizumab and Convalescent Plasma Therapy for COVID-19: A Systematic Review
ABSTRACT
Background: The current pandemic referred to as coronavirus disease 2019 (COVID-19), has identified the causative respiratory
pathogen as severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2). High viral load leads to cytokine storm (CS) and other
symptoms that leads to severity, mortality and infectivity of patients.
Methods: A review of databases PubMed and Google Scholar was conducted from May 25 to June 16, 2020. After exclusion of
systematic reviews, studies on other treatment strategies, meta-analysis and articles not related to COVID-19 treatment, 21 clinical
studies on COVID-19 infection of tocilizumab (TCZ) and convalescent plasma (CP) were identified and included.
Results: 13 TCZ studies demonstrated benefit in reducing interleukin-6 (IL-6) and C-reactive protein (CRP), thereby attenuate
CS in severe cases and 7 CP studies demonstrated benefit in decreasing viral load, IL-6 and CRP in critically ill patients. A combination
study of TCZ and CP were included which showed significant benefit of CP. A total of 151 patients were treated with TCZ and 55 were
treated with CP. A single study of both showed results similar to control group, but CP had a positive impact on infection.
Conclusion: TCZ can attenuate CS while CP can decrease viral load, acute respiratory distress syndrome (ARDS) and
inflammatory markers. CP has better benefits in comparison to TCZ and can be used in both early and severe stage of infection while
TCZ is more appropriate in the severe stage. Both drugs may be potential candidate in alleviating severity, mortality and infectivity
in COVID-19 infection.
KEYWORDS
Tocilizumab; Convalescent plasma; Interleukin-6; C-reactive protein; Cytokine storm; Viral load; COVID-19
ABBREVIATIONS:
ARDS: Acute Respiratory Distress Syndrome; CT: Computer Tomography; CP: Convalescent Plasma; COVID-19: Coronavirus Disease 2019; CRP: C - reactive protein; CS: Cytokine Storm; DRESS: Drug Reaction with Eosinophilia and Systemic Symptoms Syndrome; FDA: Food and Drug Administration; INR: International Normalized Ratio; IFN: Interferon; IL: Interleukin; IV: Intravenous; IVIG: Immunoglobulin; mIL-6R: Membrane Bound Interleukin-6 Receptor; PRISMA: Preferred Reporting Items for Systematic Review and Meta-Analysis; SC: Subcutaneous; sHLH: Secondary Hemophagocytic Lymphohistiocytosis; SARSCov- 2: Severe Acute Respiratory Syndrome Coronavirus-2; SOC: Standard of Care; sIL-6R: Soluble Interleukin-6 Receptor; TCZ: Tocilizumab; TNF: Tumor Necrosis Factor
INTRODUCTION
COVID-19 is a major respiratory illness widespread around the globe and was first reported in Wuhan, China, in December 2019. The etiological agent, SARS-CoV-2 is a single stranded RNA virus of betacoronavirus family [1]. The viral load peaks within 5 days after onset of symptoms and correlates to the various clinical manifestations observed [2]. A crucial event in COVID-19 is the excessive release of pro-inflammatory cytokines leading to an aggravated inflammatory response known as CS [3]. Released cytokines include IL-1, IL-6, IL-2, IL-7, granulocyte colony stimulating factor, interferon (IFN-γ) inducible proteins, chemoattractant protein and tumor necrosis factor (TNF-α) [4]. It is the major cause of ARDS and multiple organ failure leading to disease aggravation [5]. IL-6 is suggested to play a key role in cytokine storm [6]. IL-6 receptor has two forms, soluble (sIL- 6R) and membrane bound (mIL-6R). IL-6 binds to sIL-6R form a complex, bind to gp130 on cell membrane to complete trans-signal transduction and play a proinflammatory role [7]. TCZ, an IL-6 receptor blocker may be potential in attenuating CS [8].
CP is a passive immunization therapy to obtain instant, shortterm fortification against infectious agents by introducing a pathogen specific antibody to patients. The mechanism involved is that of antigen-antibody, where the specific antibody binds to the specific antigen and block its interaction with cell receptor, which is extremely applicable in the case of viral disease [9]. In the absence of specific antiviral therapy, TCZ and CP can be considered for treatment of severe patients that may provide symptomatic relief and decrease the severity and mortality of infection.
METHODS
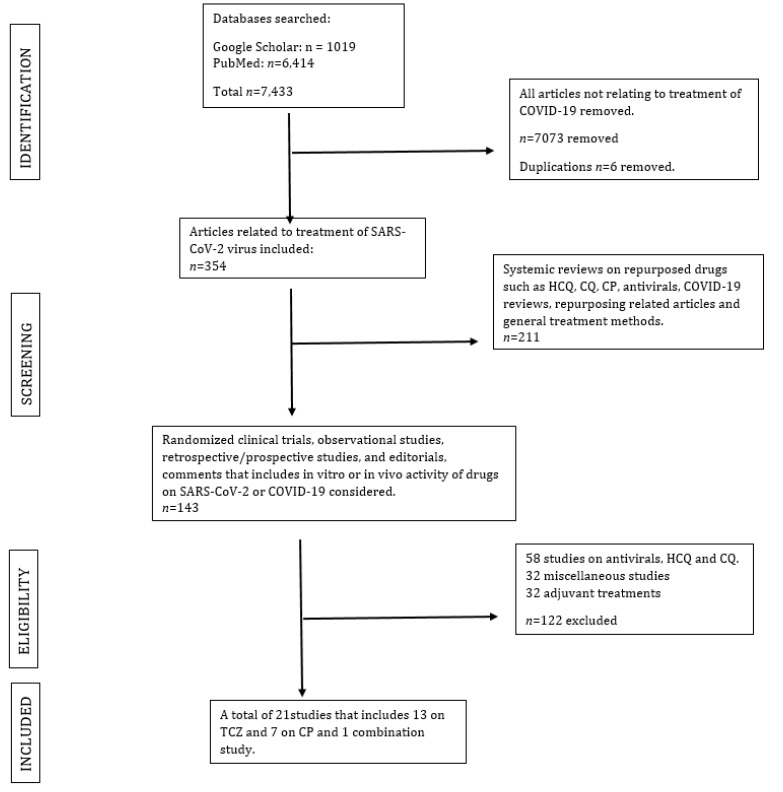
The systematic review was conducted according to the Preferred Reporting Items for Systematic Review and Meta- Analyses (PRISMA) guidelines (flow diagram- Figure 1). There is no protocol for this review. From May 25 2020 to June 16 2020, a literature review conducted in databases PubMed and Google Scholar with the term “treatment of COVID-19”.Search yielded a total of 7433 articles and 6 duplicated articles were removed followed by the exclusion of articles not related to the treatment of COVID-19. The resulting amount of 354 articles assessed on the basis of abstract for differentiating clinical in vitro, in vivo studies and hypothesis literature on SARS-CoV-2 treatments. Letters to editors, comments or pre-prints considered if it contains a clinical study with relevant results. Out of 143 articles identified, 122 excluded as it does not correlate to TCZ or CP therapy. Remaining 21 articles were downloaded and reviewed in detail. The number of participants, disease progression and descriptive summary of outcomes obtained from the studies.
RESULTS
The systematic review identified 13 studies of TCZ that includes 5 retrospective, 5 case reports, 3 case series and 7 CP studies that includes 3 prospective, 1 retrospective, 2 case reports and 1 case series. An observational study which used both TCZ and CP were included. A total of 151 patients were treated with TCZ and 55 were treated with CP.
TCZ Therapy
A retrospective study of 21 severe COVID-19 patients received TCZ, out of which 3 patients had a second dose due to fever. The median levels of IL-6 in 18 patients were 153.44 ± 296.3 pg/mL and remaining 3 patients had 6.95, 7.4 and 7.72% IL-6 cells in peripheral blood. Fever was reduced on the first day after therapy. Within next 5 days, 75% had lowered oxygen supplementation. Although the values of IL-6 did not decrease significantly, 90% had their lung opacities absorbed, 52.6% had lymphocytes normalized and CRP decreased significantly in 84.2% [10].
In another retrospective study, 15 COVID-19 patients were treated with TCZ. The CRP levels dropped from 126.9mg/L to 11.2 mg/L by the end of therapy. A single dose TCZ given to the 4 critically ill patients had 3 deaths. The IL-6 levels after the TCZ therapy in 10 patients showed a short spike and then returned to normal whereas in 4 critically ill patient with failed therapy, increased IL-6 was found [11]. A retrospective analysis of 25 severe COVID-19 patients treated with multiple doses of TCZ had their outcomes assessed in 14 days of therapy.
Post TCZ administration, the temperature normalized. By day 7, median CRP declined from 193mg/L to 7.9mg/L and significant radiological improvement in 44% observed. By day 14, 68% showed radiological improvement and 9 patients discharged. 92% patients experienced at least one adverse event which included anemia (64%), elevated alanine aminotransferase (44%) and QT interval prolongation (20%). [12]. A retrospective trial on 65 severe COVID-19 patients had 32 in TCZ and 33 in control with hyper inflammatory features and was treated with intravenous (IV) TCZ (400mg). The primary end point was improvement in the clinical status assessed over a period of 28 days. Death and discharge were 16% and 63% in TCZ and 33% and 49% in the control. The median time of discharge in TCZ was 13.5 and control was 14 days. Clinical improvement achieved in 69% in TCZ and 61% in the control. Serious adverse events recorded 25% in TCZ and 27% in control, which includes bacteremia (13% and 12%), pulmonary thrombosis (6% and 9%), increased serum transaminase (15% and 18%) and transitory neutropenia (16% in TCZ only) [13]. In another retrospective analysis treated with IV TCZ (8mg/kg) matched using propensity score to patients treated with standard of care (SOC) who were prospectively enrolled in the SMAtteo COvid-19 Registry (SMACORE). 42 patients analysed after propensity score matching. The effect of TCZ on mortality in 7 days was not significant. Interaction between time and treatment were significant for international normalized ratio (INR) and CRP [14].
A 63-year-old COVID-19 patient had fever, oxygen saturation at 93% and bilateral ground glass opacities on computer tomography (CT). On day 4 the oxygen saturation was 89% with increased levels of IL-6 and CRP. A single dose of intravenous TCZ, at 8mg/ kg was given. Oxygenation improved, IL-6 declined after reaching its peak and CRP decreased significantly. Lymphocyte count increased significantly. On day 13 chest CT confirmed significant improvement. Mild liver impairment on day 10 and leucopenia on day 16 noted, which was self-resolved [15]. In a case report of a 42-year-old cancer patient affected with COVID-19, had fever and bilateral ground-glass opacities on CT. Antiviral therapy given and on day 8, sudden dyspnea and drop in oxygen saturation occurred. Oxygen supplementation with IV TCZ, 8 mg/kg, 8h apart provided. Chest CT confirmed improvement and CRP decreased from 225 mg/L to 33 mg/L in 4 days. Patient fully recovered [16]. A case report of 57-year-old male patient presented with chest tightness, fever and progressed to shortness of breath with oxygen saturation 90% and CT with extension of ground glass opacities. Laboratory test showed IL-6 and CRP increased. Patient given IV TCZ 400mg. Initially IL-6 increased, CRP, white blood cells and neutrophils reduced. Later the IL-6 reduced remarkably [17
Another case report of a 25-year-old male had fever, oxygen saturation at 90% with increased CRP, D-dimer and ferritin. IV TCZ (8mg/Kg body weight) was administered. Initially CRP, ferritin, D-dimer and IL-6 showed increase but within 5 days post therapy, the following laboratory parameters returned to normal. Chest X-ray showed complete resolution of abnormalities followed by discharged [18]. A 70-year-old COVID-19 patient progressed to leucocytopenia, lymphocytopenia and was administered with IV TCZ 600 mg. Patient developed an itching generalized cutaneous “toxic erythema-like rash” and severe eosinophilia. Later COVID-19 symptoms decreased, but eosinophil count increased and the skin rash was persistant. A differential diagnosis, drug reaction with eosinophilia and systemic symptoms syndrome (DRESS) was considered, but the rapid onset and absence of organ involvement was contrary to the disease [19].
In a series of cases 3 COVID-19 pneumonia patients, treated with subcutaneous (SC) TCZ, resulted in IL-6 levels decreased from 106.1pg/mL to 6.32 pg/mL in patient 1, 72.65pg/mL to 5.55 pg/mL in patient 2 and 64.3pg/mL to 40.5 pg/mL in patient 3. All patients demonstrated improvement in radiological findings, fever and oxygen demand [20]. Another case series of 3 COVID-19 patients with rapidly progressing respiratory insufficiency were administered with multiple doses of TCZ. A reduction of CRP was seen in patient 2 from 151 to 13 mg/L, patient 3 from 250 to 92 mg/L and normalized in patient 1. Also, clinical recovery with improvement in CT and respiratory functions were observed in all patients [21]. In a case study of 2 COVID-19 patients treated with IV TCZ resulted in secondary hemophagocytic lymphohistiocytosis (sHLH). Patient 1 developed (sHLH) despite the decrease in CRP whereas patient 2 demonstrated continuous rise of inflammatory markers and expired [22].
CP Therapy
The clinical study had 5 critically ill COVID-19 patients with ARDS treated by 2 consecutive transfusion of 200 to 250 mL CP. In polymerase chain reaction, higher value of Ct is correlated with lower viral load. Post transfusion, Ct increased from 22 to 35.9 which implies lowering of viral load. Body temperature normalized in 4 patients within 3 days following improvement in oxygenation, SARS-CoV-2 negative and ARDS resolved [23]. Another study had 10 severe COVID-19 patients treated with a single dose of 200 mL of CP transfused into the patients. Post transfusion, all symptoms improved within 1 to 3 days, inflammatory markers reduced and absorption occurred in CT. 7 out of 10 patients had improved lymphocyte count. By day 6, all patients had undetectable SARSCoV- 2. A control group of 10 patients were compared. 3 discharged and 7 improved in CP whereas 3 deaths, 6 stabilized and 1 improved in control [24]. A retrospective study of 21critically ill COVID-19 patients had 6 treated with CP and 15 as control. The median volume of plasma infused was 300 mL. Death occurred in 5 patients in CP group and 14 in control. Also, all 6 patients achieved viral clearance after CP transfusion. The survival period was longer in the treatment group compared to control [25]. The following clinical study enrolled 25 severe or life threatening COVID-19 patients. The primary endpoint is the safety of CP therapy and the secondary endpoint was clinical improvement at day 14. At day 7, post transfusion, 36% improved from baseline, 52% no change, 3 deteriorated and 28 % discharged. By day 14, 76% improved from baseline and an additional 4 patients discharged. The values of CRP decreased [26].
In a case report of a 100-year-old patient with slight patchy and cord like dense enhancements on CT and high viral load was treated with 2 doses of 200 mL CP followed by 100 mL, 4 days later. Lymphocyte count increased, IL-6, CRP and viral load decreased with improved clinical outcomes [27]. Another case report of a 50-yearold COVID-19 patient had leucopenia and lymphocytopenia. Oxygen saturation declined and progressed to acute severe hypoxemic respiratory failure. Further the Lymphocytes declined with multiple ground glass opacities and high-density consolidations on CT. Patient treated with 3 consecutive session of CP (6000 mL) followed by IVIG 20g. The oxygenation saturation of patient improved along with clinical symptoms. After a fourth transfusion followed by IVIG, patient made prompt recovery. All the symptoms alleviated, CT showed improvement and patient discharged [28].
A case series had 4 COVID-19 patients treated with CP. 2 patients had decreasing oxygen saturation progressing to severe condition and the other 2 had ARDS progressing to critical stage. Administration of CP resulted in adsorption in CT with decreased viral load and improved clinical symptoms in all patients [29].
Comparison of CP and TCZ
The observational study had 6 severe COVID-19 patients in which 3 were treated with CP and 3 with TCZ. It was observed that CRP and IL-6 levels were significantly reduced and the lymphocyte and prothrombin time were improved in patients treated with CP. The reduction of inflammatory markers had no significance in TCZ treated patients [30].
DISCUSSION
Recent studies have established that CS or sHLH leads to ARDS and multi-organ failure, thereby increase the severity and mortality of COVID-19 infection [31]. Similarly, high viral load was identified to be directly proportional to severity and mortality of infection.
The following review identified that TCZ therapy is beneficial in symptomatic improvement of severe COVID-19 patients, [10- 13,15-22,30] except in a single study where TCZ administration had similar results as that of the control. Similarly, CP studies demonstrated a broad range of treatment benefits and achieved clinical improvements [23,24,26-29]. Although CP had results identical to control, it was able to attain viral clearance in all patients with longer survival period [25].
TCZ demonstrated reduction of IL-6 [10,11,15,17,18,20] in most studies. IL-6, a pro-inflammatory cytokine is found to be a decisive part of CS, therefore the use of IL-6 receptor blocker TCZ, approved by Food and Drug Administration (FDA) for CS, [32] is probably the right choice in patients with elevated IL-6. In a particular study where 4 critically ill patients had failed TCZ therapy demonstrated a high rise in IL-6 and CRP [11] which further proves that IL-6 have a potential role in CS that leads to severity and mortality of patients. In some of the studies the IL-6 had an initial rise before decreasing [11,15,17,18] which may be attribute to the fact that TCZ binds to the IL-6 receptors [33] thereby IL-6 levels rise due to unavailability of receptors for binding. The review elucidated that TCZ therapy in COVID-19 reduced CRP [10-12,14-18,20-22] levels. A significant reduction was identified in studies [10,14,15]. CRP is a pentameric protein synthesized by the hepatocytes and their levels are directly proportional to inflammatory responses. It is primarily induced by IL-6 during the acute phase of inflammation [34] which correlates to the studies analysed. Reduced IL-6 was associated with decreased CRP.
Viral clearance was achieved by CP transfusion in multiple studies [23,25,27,29] as the potential mechanism of CP in SARSCoV- 2 is viral neutralization. Other possible mechanism such as antibody-dependent cellular cytotoxicity and/or phagocytosis are also involved [35]. Lack of delayed viral clearance may be due to concomitant administration of glucocorticoids, [24] which is established in the literature. [36]. The CP therapy also led to reduced inflammatory markers, [24,30] decreased CRP and [26,27,30]. IL-6. Since high viral load is correlated to CS and therefore escalated severity and mortality in COVID-19 infection, [37] the reduction of viral load can attenuate hyperinflammation, thereby reduce respiratory problems inflammatory markers. Also, the use of concomitant steroids may have played a role in reduction of inflammatory markers [24,26] but the possibility of CP to reduce inflammation [27] and resolve ARDS were also observed [23,29].
The D-dimer, ferritin [18] and INR [14] was reduced by TCZ administration and a significant reduction of prothrombin time was observed in CP therapy, (30) which points to the fact that both therapy may prevent hypercoagulation. Study showed that patients had inflammatory thrombosis which indicates that increased pro-inflammatory cytokines and CRP leads to increased D-dimer, ferritin and fibrinogen levels [38]. Similarly, high viral load leads to increased inflammatory cytokines [39]. Therefore, the reduction of viral load and prevention of CS can possibly eliminate occurrence of hypercoagulation. Also, an improvement in lymphocytes were noted in TCZ (10, 15) and CP (24, 27, 30) therapy which can be attributed to clearance of pro-inflammatory cytokines and SARSCoV- 2 [40].
Fever was reduced [10,20] along with radiological improvement [10,12,15,18,21] and better oxygenation [10,15,18,20] in patients. Improvements in oxygenation, [23,28] reduced fever [23] and CT adsorption [24,29] were also observed post CP therapy. The CP may also replenish other plasma constituents and exert beneficial effects by substituting reduced components in patients [40]. A study has pointed out fever, as the hallmark of CS which is induced by IL-6 and TNF presence. CS also increases the capillary permeability and results in the leakage of proteins leading to organ damage and injury [41]. Lowered viral clearance may increase hyperinflammation [42]. Thus, the resolution of fever, adsorption in CT and better oxygenation is related to resolving or decreasing cytokine storm or increasing viral clearance which were found in all studies.
The TCZ dose-response curve flattens at higher exposure resulting in smaller efficacy gains for incremental doses,[33] therefore instead of single high dose, multiple doses should be given which is implied in a study where single dose of TCZ did not provide positive therapeutic outcomes in critically ill patients [11]. Despite TCZ therapy sHLH developed in 2 patients. One patient had CRP reduction, other patient expired with rising inflammatory markers [22] and lack of response in 4 critically ill patients might correlate to the fact that administration of TCZ at early stage of severe infection may be more beneficial in CS.
CP is used to treat severe or life-threatening COVID-19 infection after submitting online form by doctors and obtaining a verbal approval from FDA [43]. As a result, the CP therapy is used only in the very late stage of infection, which can probably be a disadvantage. Although CP can be used for treating infection at varying level of severity of infection, its activity is most effective when administered prophylactically or early after the onset of symptoms, as the inoculum is small and easier to tackle.
Adverse reactions such as anemia, elevated transaminase level, QT prolongation occurred in one study. Another study had adverse reactions between TCZ and control which includes bacteremia (13% and 12%), pulmonary thrombosis (6% and 9%), increased serum transaminase (15% and 18%) and transitory neutropenia (16% in TCZ only). Mild liver impairment and leucopenia observed, which was self-resolved. A generalized, itching cutaneous “toxic erythema-like rash” and severe eosinophilia was observed in another. The following adverse effects can be attribute to TCZ or concomitant drugs used or consequence of SARS-CoV-2 infection. Mild elevation of transaminase levels, neutropenia and skin infections are TCZ associated adverse reactions. Also, liver impairment occurs mainly when TCZ is administered with concomitant hepatotoxic drug, [44,45] such as chloroquine. Also, hypereosinophilia and erythroderma are documented to be TCZ induced [46,47]. Meanwhile the QT interval prolongation is associated with hydroxychloroquine and azithromycin treatment, which is well documented in COVID-19 patients [48]. Pulmonary thrombosis occurrence is a hallmark of COVID-19 [49] and bacteremia may not be prevalent among COVID-19 patients but can occur [50]. Also, the SC TCZ administration may be able to reduce the dose but may increase skin infection at the injection site [45].
1 patient had morbilliform rash, 2 patient developed deep vein thrombosis (DVT) and 1 patient developed a DVT and pulmonary embolism, the following thrombotic complication is well associated with COVID-19 infection. No adverse reactions were noted in any of the patients treated with CP. Potential risk involved are transfusiontransmissible diseases, allergic transfusion reactions (serum sickness, allergy to serum constituents), transfusion associated circulatory overload, transfusion related acute injury, susceptibility to subsequent infection and antibody dependent enhancement.
A comparison study of TCZ and CP, suggested that CP reduced inflammatory markers significantly than TCZ in severe COVID-19. (30) The main advantage of CP is that it can be easily available and cost effective but acquiring necessary quantity is a problem [51]. Pertaining to the current circumstances of large group of people with cure rate of 98.6%, [52] obtaining necessary quantity of CP might be possible. TCZ have the higher chance of adverse events occurrence but is easily available. The review identifies that CP can decrease viral load, attenuate CS and improve clinical symptoms. On the other hand, TCZ is specific in blocking IL-6 and CRP that is involved in CS and thereby attain symptomatic improvement. Therefore, TCZ can be used in later stage where elevation of IL-6 observed and CP can be used in both later and early stage where latter administration has better benefits.
Limitations of the studies include retrospective design [12,13] small cohort [13,14,23,26] and administration of concomitant medications that made it difficult to properly attribute the effectiveness to the specific drug [12,15,14,23]. The following limitations can be attributed to the dire circumstances of the pandemic and severity of the infections. Also, the amount of literature available is low for both drugs which implies the necessity of larger, more randomized controlled trials.
CONCLUSION
TCZ attenuate CS and CP decrease viral load and both can prevent hypercoagulation, improve lymphocyte count and other clinical symptoms. Additionally, CP may also decrease inflammatory markers including IL-6 and CRP by viral clearance and has a high chance of decreasing CS. TCZ has a higher possibility of adverse drug reaction occurrence and is more of an adjuvant drug. TCZ may be used in the early phase of severe stage and CP may be used in both early and late stage of COVID-19 infection. Thus, CP has a better advantage over TCZ but both may be potential candidate in COVID-19 infection in reducing severity, infectivity and mortality.
REFERENCES
- Rothan HA, Byrareddy SN (2020) The epidemiology and pathogenesis of coronavirus disease (COVID-19) outbreak. J Autoimmun 109: 102433.
- Wölfel R, Corman VM, Guggemos W (2020) Virological assessment of hospitalized patients with COVID-2019. Nature 581(7809): 465-469.
- Ragab D, Salah Eldin H, Taeimah M, Khattab R, Salem R (2020) The COVID-19 cytokine storm; what we know so far. Front Immunol 11: 1446.
- Zhang W, Zhao Y, Zhang F (2020) The use of anti-inflammatory drugs in the treatment of people with severe coronavirus disease 2019 (COVID-19): The Perspectives of clinical immunologists from China. Clin Immunol 214: 108393.
- Ye Q, Wang B, Mao J (2020) The pathogenesis and treatment of the `Cytokine Storm’ in COVID-19. J Infect 80(6): 607-613.
- Zhou Y, Fu B, Zheng X, Wang D, Zhao C, et al. (2020) Pathogenic T-cells and inflammatory monocytes incite inflammatory storms in severe COVID-19 patients. Natl Sci Rev 7(6): 998-1002.
- Wolf J, Rose-John S, Garbers C (2014) Interleukin-6 and its receptors: a highly regulated and dynamic system. Cytokine 70(1): 11-20.
- Zhang C, Wu Z, Li JW, Zhao H, Wang GQ (2020) Cytokine release syndrome in severe COVID-19: interleukin-6 receptor antagonist tocilizumab may be the key to reduce mortality. Int J Antimicrob Agents 55(5): 105954.
- Alzoughool F, Alanagreh L (2020) Coronavirus drugs: Using plasma from recovered patients as a treatment for COVID-19. Int J Risk Saf Med 31(2): 47-51.
- Xu X, Han M, Li T (2020) Effective treatment of severe COVID-19 patients with tocilizumab. Proc Natl Acad Sci 117(20): 10970-10975.
- Luo P, Liu Y, Qiu L, Liu X, Liu D, et al. (2020) Tocilizumab treatment in COVID-19: A single center experience. J Med Virol 92(7): 814-818.
- Alattar R, Ibrahim TBH, Shaar SH (2019) Tocilizumab for the treatment of severe coronavirus disease 2019. J Med Virol.
- Campochiaro C, Della-Torre E, Cavalli G (2020) Efficacy and safety of tocilizumab in severe COVID-19 patients: a single-centre retrospective cohort study. Eur J Intern Med 76: 43-49.
- Colaneri M, Bogliolo L, Valsecchi P (2020) Tocilizumab for treatment of severe COVID-19 patients: preliminary results from Smatter COVID19 Registry (SMACORE). Microorganisms 8(5): 695.
- Xu CY, Lu SD, Ye X (2020) Combined treatment of tocilizumab and chloroquine on severe COVID-19: A case report. An International Journal of Medicine 569-572.
- Michot JM, Albiges L, Chaput N (2020) Tocilizumab, an anti-IL-6 receptor antibody, to treat COVID-19-related respiratory failure: a case report. Ann Oncol 31(7): 961-964.
- Wang L, Peng X, Wang ZH, Cai J, Zhou FC (2020) Tocilizumab in the treatment of a critical COVID-19 patient: a case report. Eur Rev Med Pharmacol Sci 24(10): 5783-5787.
- Van Kraaij TD, Mostard RL, Ramiro S (2020) Tocilizumab in severe COVID-19 pneumonia and concomitant cytokine release syndrome. Eur J Case Rep Intern Med 7(5):001675.
- Sernicola A, Carnicelli G, Di Fraia M (2020) Toxic erythema and eosinophilia associated with tocilizumab therapy in a COVID-19 patient. J Eur Acad Dermatol Venereol 34(8): e368-370.
- Mazzitelli M, Arrighi E, Serapide F (2020) Use of subcutaneous tocilizumab in patients with COVID-19 pneumonia. J Med Virol.
- Di Giambenedetto S, Ciccullo A, Borghetti A (2020) Off-label use of tocilizumab in patients with SARS-CoV-2 infection. J Med Virol.
- Radbel J, Narayanan N, Bhatt PJ (2020) Use of tocilizumab for COVID- 19-Induced cytokine release syndrome: A cautionary case report. Chest 158(1): e15-e19.
- Shen C, Wang Z, Zhao F (2020) Treatment of 5 critically ill patients with COVID-19 with convalescent plasma. JAMA 323(16):1582-1589.
- Duan K, Liu B, Li C (2020) Effectiveness of convalescent plasma therapy in severe COVID-19 patients. Proc Natl Acad Sci 117(17): 9490-9496.
- Zeng QL, Yu ZJ, Gou JJ (2020) Effect of convalescent plasma therapy on viral shedding and survival in patients with coronavirus disease 2019. J Infect Dis 222(1): 38-43.
- Salazar E, Perez KK, Ashraf M (2020) Treatment of coronavirus disease 2019 (COVID-19) Patients with convalescent plasma. Am J Pathol 190(8): 1680-1690.
- Kong Y, Cai C, Ling L (2020) Successful treatment of a centenarian with coronavirus disease 2019 (COVID-19) using convalescent plasma. Transfus Apher Sci 102820.
- Shi H, Zhou C, He P (2020) Successful treatment of plasma exchange followed by intravenous immunogloblin in a critically ill patient with 2019 novel coronavirus infection. Int J Antimicrob Agents. 105974.
- Zhang B, Liu S, Tan T (2020) Treatment with convalescent plasma for critically ill patients with severe acute respiratory syndrome coronavirus 2 infection. Chest 158(1): e9-e13.
- Luo S, Yang L, Wang C, Liu C, Li D (2020) Clinical observation of 6 severe COVID-19 patients treated with plasma exchange or tocilizumab. Zhejiang Da Xue Xue Bao Yi Xue Ban 49(2): 227-231.
- Soy M, Keser G, Atagündüz P, Tabak F, Atagündüz I, Kayhan S (2020) Cytokine storm in COVID-19: pathogenesis and overview of antiinflammatory agents used in treatment. Clin Rheumatol 39(7): 2085- 2094.
- Zhang S, Li L, Shen A, Chen Y, Qi Z (2020) Rational use of tocilizumab in the treatment of novel coronavirus pneumonia. Clin Drug Investig 40(6): 511-518.
- Alten R (2011) Tocilizumab: a novel humanized anti-interleukin 6 receptor antibody for the treatment of patients with rheumatoid arthritis. Ther Adv Musculoskelet Dis 3(3): 133-149.
- Nehring SM, Goyal A, Bansal P, Patel BC (2020) C Reactive Protein (CRP). In: Stat Pearls. Treasure Island, Stat Pearls Publishing, Florida, USA.
- Casadevall A, Pirofski LA (2020) The convalescent sera option for containing COVID-19. J Clin Invest 130(4): 1545-1548.
- Ling Y, Xu SB, Lin YX (2020) Persistence and clearance of viral RNA in 2019 novel coronavirus disease rehabilitation patients. Chin Med J (Engl) 133(9): 1039-1043.
- Rajpal A, Rahimi L, Ismail-Beigi F (2020) Factors leading to high morbidity and mortality of COVID-19 in patients with type 2 diabetes. J Diabetes.
- Mouhamed Yazan Abou-Ismail, Akiva D, Sargam K, Yasmin A, Lalitha N (2020) The hypercoagulable state in COVID-19: Incidence, pathophysiology, and management. Thrombosis Research 194: 101-115.
- Urra JM, Cabrera CM, Porras L, Ródenas I (2020) Selective CD8 cell reduction by SARS-CoV-2 is associated with a worse prognosis and systemic inflammation in COVID-19 patients. Clin Immunol 217: 108486.
- Roback JD, Guarner J (2020) Convalescent plasma to treat COVID-19: Possibilities and challenges. JAMA.
- Mangalmurti N, Hunter CA (2020) Cytokine Storms: Understanding COVID-19. Immunity 53(1):19-25.
- Soy M, Keser G, Atagündüz P, Tabak F, Atagündüz I, et al. (2020) Cytokine storm in COVID-19: pathogenesis and overview of anti-inflammatory agents used in treatment. Clin Rheumatol 39(7): 2085-2094.
- Tanne JH (2020) Covid-19: FDA approves use of convalescent plasma to treat critically ill patients. BMJ 368: m1256.
- Bloch EM, Shoham S, Casadevall A (2020) Deployment of convalescent plasma for the prevention and treatment of COVID-19. J Clin Invest 130(6): 2757-2765.
- Sheppard M, Laskou F, Stapleton PP, Hadavi S, Dasgupta B (2020) Tocilizumab (Actemra). Hum Vaccin Immunother 13(9): 1972-1988.
- Morrisroe K, Wong M (2020) Drug-induced hypereosinophilia related to tocilizumab therapy for rheumatoid arthritis. Rheumatology (Oxford) 54(11): 2113-2114.
- Nakamura M, Tokura Y (2009) Tocilizumab-induced erythroderma. Eur J Dermatol 19(3): 273-274.
- Chorin E, Wadhwani L, Magnani S (2020) QT interval prolongation and torsade de pointes in patients with COVID-19 treated with hydroxychloroquine/azithromycin. Heart Rhythm 17(9): 1425-1433.
- Cattaneo M, Bertinato EM, Birocchi S (2020) Pulmonary embolism or pulmonary thrombosis in COVID-19? is the recommendation to use high-dose heparin for thromboprophylaxis justified. Thromb Haemost 120 (8): 1230-1232.
- Sepulveda J, Westblade LF, Whittier S (2020) Bacteremia and blood culture utilization during COVID-19 surge in New York City. J Clin Microbiol 58(8): e00875-e00920.
- Abolghasemi H, Eshghi P, Cheraghali AM (2020) Clinical efficacy of convalescent plasma for treatment of COVID-19 infections: Results of a multicenter clinical study. Transfus Apher Sci 102875.
- (2020) Coronavirus (COVID-19) mortality rate. Worldometer.
Article Type
Research Article
Publication history
Received date: August 28, 2020
Published date: September 08, 2020
Address for correspondence
Steffi Rose Mathew, Pharm D, Qatar
Copyright
©2020 Open Access Journal of Biomedical Science, All rights reserved. No part of this content may be reproduced or transmitted in any form or by any means as per the standard guidelines of fair use. Open Access Journal of Biomedical Science is licensed under a Creative Commons Attribution 4.0 International License
How to cite this article
Steffi RM. Comparison of Tocilizumab and Convalescent Plasma Therapy for COVID-19: A Systematic Review. 2020 - 2(5) OAJBS.ID.000215.